Biological Phosphorus Elimination
Biological phosphorus elimination is getting more and more important in the recovery of phosphorus as a limited resource and for compliance with statutory limit values. With VIT®, the Bio-P process can be monitored easily.
Phosphorus is vital
Nitrogen and phosphorus are the most important nutrient elements for all life on earth. Without phosphorus, no living organisms could exist. Phosphorus constitutes 0.099 % of the earth crust and cannot be substituted by other elements. In contrast to nitrogen, it is not part of the atmosphere. In every cell, whether bacterial, plant or animal cell, phosphorus serves as central element of the ATP (adenosine triphosphate) molecule – the universal currency of life. In the course of nutrient degradation, energy is created and temporarily stored in the form of ATP. Subsequently, ATP can be transported to other locations in the cell where it is required. For this reason, an organism could not function without phosphorus. Yet phosphorus is also needed in many other physiological processes: it is, for example, an essential factor in bone formation in humans and is an important part of the genetic material in all forms of life.
The fight for phosphorus
Phosphorus was discovered in 1669 and has been put to industrial use as an integral component of matchsticks. Today, 80 % of all phosphates recovered worldwide are used in fertilizer production, 12 % are applied in the production of detergents, 5 % in animal feed production, while other applications account for 3 %. Ca. 160 millions of tons of phosphate are mined each year from natural deposits. Those reservoirs are for the most part concentrated in 4 countries which are in possession of 80% of the natural deposits: Morocco, China, South Africa and Jordan. Key players in phosphorus mining are above all China, USA, Morocco, Russia and Tunisia. Europe has to cover 90% of its requirements with imports from these countries.
In its white paper "A Sustainable Global Society", which was published in 2011, The Chemical Sciences and Society Summit (CS3) stated that natural phosphorus deposits will probably be depleted in the next 30 - 100 years. As a few countries are almost in complete control of a resource which is required by all others, conflicts are inevitable. A limitation of phosphorus touches the national security of numerous nations, as even today, the production of crop plants in many regions is at risk due to the limited occurrence of phosphorus.
THE DISCOVERY OF PHOSPHORUS
The German pharmacist and alchemist Hennig Brand is credited with discovering phosphorus. He discovered it while attempting to find the philosopher’s stone. The philosopher’s stone was said to be able to transform base metals into gold and silver. Brand heated urin and obtained a white substance which glowed in the dark and was flammable. Due to the white flame it produced while burning, Brand named it “cold fire”.
DISCOVERY OF NEW PHOSPHORUS DEPOSITS
In 2012 new phosphorus deposits were discovered in Northern Africa and Iraq. According to estimate by the German Federal Government, this could mean that the need for phosphorus is covered for about the next 400 years after all.
The need for phosphorus recovery
Given the fact that natural reservoirs of phosphorus are nearly exhausted, a recycling of phosphorus is mandatory. Very large amounts of phosphorus are accumulated in waste water: ca. 70% originate from human and animal feces and the remaining 30% from cleaning and washing agents and kitchen waste. For this reason, most technical solutions focus on the recovery of phosphorus from sewage sludge produced by wastewater treatment plants.
The dilemma: good phosphorus, bad phosphorus
On the one hand, phosphorus is a limited resource and has to be recovered if the supply of food for a steadily growing human population is to be ensured. On the other hand, phosphorus that enters wastewater treatment plants with incoming sewage poses a problem. Total emissions in Germany have decreased by 70% since the 1980ies, but many water bodies are heavily contaminated even today. In wastewater, phosphorus is largely present in its inorganic form (orthophosphate) and, in lower amounts, as organic phosphorus in non-dissolved and dissolved form. If phosphorus leaves the wastewater treatment plant and enters the recipient, eutrophication of the receiving water body can easily occur. Frequently, phosphorus is the limiting factor for microorganisms and plants. If this limitation is removed, an almost unchecked proliferation of microorganisms and plants can result. This leads to increased consumption of oxygen and thus to the development of anaerobic zones. The result is eutrophication of the water body and the death of resident fish.

New, more stringent limit values
In order to avoid eutrophication of water bodies, the German Federal Government has commited to a further reduction of phosphorus emissions in the course of its sustainability strategy. This means that wastewater treatment plant operators are coming under increased pressure to meet the limit values if they want to avoid having to pay increased charges.
Chemical and biological phosphate removal
In general, there are two alternatives for phosphate removal. The first option is chemical precipitation of phosphates by use of metallic salts. In the aeration tank, ferric and aluminium metal salts form insoluble chemical compounds with previously dissolved phosphates. These metallic phosphates can subsequently be removed together with the excess sludge at the second clarification stage. As a second option, phosphates can be absorbed by microorganisms, which are also removed together with the excess sludge. As a result, the phosphorus present in the excess sludge is bound either biologically (within the microorganisms) or chemically in the form of metallic phosphates.
The natural potential of biological phosphorus removal
The bacteria referred to as “Bio-P microorganisms” are specialized polyphosphate-accumulating microorganisms (PAO) that are able to store phosphate in their cells under aerobic conditions and in doing so, remove it biologically from the wastewater. They are in direct competition with glycogen-accumulating microorganisms (GAO), which are competing with them for the same carbon sources under anoxic conditions. Every wastewater treatment plant has a natural potential for biological phosphate removal. As a result, ca. 35% of the phosphorus is deposited in sludge. This share can vary depending on the specific conditions in the plant. Consequently, there is an enormous natural potential which helps with reducing the phosphorus load.
SYNONYMS
Biological phosphate elimination is also known as biological phosphorus elimination, biological phosphate removal or Bio-P.
Bio-P for an enhanced biological phosphate removal
In case the wastewater treatment plant’s natural potential is not sufficient to meet the required limit values, biological phosphate removal can be enhanced by a specialized process. It includes the generation of unventilated and ventilated zones, while the microorganisms are subsequently passing through these zones. In this way, they are induced to accumulate additional amounts of phosphate in their cells, which serves as their energy storage. This is a natural safety mechanism for PAO, supposed to provide them with sufficient amounts of energy even in adverse conditions. As a result, this leads to an increased removal of phosphorus.
Control of PAO/GAO
With the VIT® gene probe technology and the product VIT® PAO/GAO or the lab service VIT® PAO/GAO, the populations of polyphosphate-accumulating microorganisms (PAO) in wastewater treatment plants can be examined. They allow a very quick assessment of a wastewater treatment plant’s basic potential for biological phosphorus removal. In addition, the ratio of PAO to the competing glycogen-accumulating microorganisms (GAO) can be monitored, allowing to systematically increase the efficiency of biological phosphate removal by targeting the process. The method can be applied directly on site or can be ordered as a lab service.
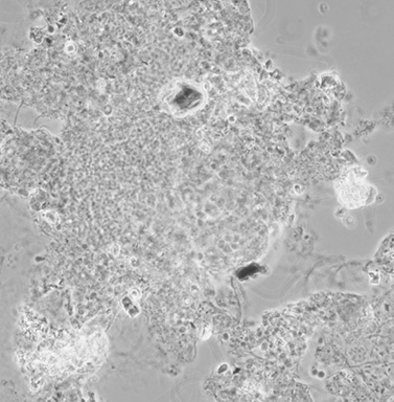
Phase contrast microscopic image of a wastewater sample.
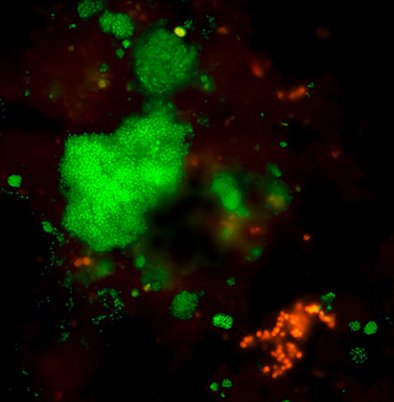
The same image after analysis with VIT®: PAO shine in red, GAO shine in green.
Options of recovery
The phosphorus bound in the excess sludge allows for a subsequent recovery of the vital chemical element. Due to its pollutant load, a direct utilization of the sludge is not recommended and in fact illegal in many countries.
There are numerous methods of phosphorus recovery. In general, there are three different groups: Firstly, the recovery from sludge mono ashes, which is quite expensive, though. Secondly, direct recycling from sludge and thirdly, recycling from the process water of sludge treatment. As up 90% of the incoming P loads are bound in sludge and the process waters contain only up to 25 % of the incoming loads, recovery from sludge via chemical extractions or crystallization is the method of choice.
Recovery allows to return the phosphorus to the cycle, because in contrast to carbon or nitrogen, phosphorus cannot be removed from wastewater via gaseous phase.

The fight for phosphorus has already begun
Phosphorus is one of the most important resources and, moreover, only limited quantities are at humanity’s disposal. If it is not recovered, it becomes lost to the phosphorus cycle. The fight for this limited resource has already started. Europe has to reduce its dependence on the few phosphorus-producing countries by increased recycling. Furthermore, the discharge of treated wastewaters still containing phosphates to the recipients leads to eutrophication of water bodies. While the situation has improved in recent years, limit values are going to be even more ambitious in the near future, which will make it very difficult for wastewater treatment plant operators to comply with the limit values. For wastewater treatment plants, VIT® PAO/GAO analytics are an important tool for the supervision of biological phosphate removal and they allow to monitor those microorganisms that are essential players in biological phosphate removal.
Avoiding contamination of water bodies with phosphate and recycling of this resource should go hand in hand. For only if phosphorus is removed from wastewater, it becomes available again for recovery in the form of excess sludge.
Related links:
The Phosphorus cycle (English): https://en.wikipedia.org/wiki/Phosphorus_cycle
Am Phosphor hängt das Schicksal der Menschheit (German): https://www.welt.de/dieweltbewegen/article13585089/Am-Phosphor-haengt-das-Schicksal-der-Menschheit.html
Der Stein des Lichtes - Elementargeschichte des Phosphors (German): http://www.spektrum.de/magazin/der-stein-des-lichtes-elementargeschichte-des-phosphors/822157